-

Understanding disease through genetics
The lab aims to uncover the mechanisms that lead to Alzheimer's disease and other neurodegenerative diseases through genetic discoveries.
-
Team science for clinical impact
Our team seeks to discover therapeutic targets and novel biomarkers for Alzheimer's disease by applying systems-level analyses and experimental validation.
-
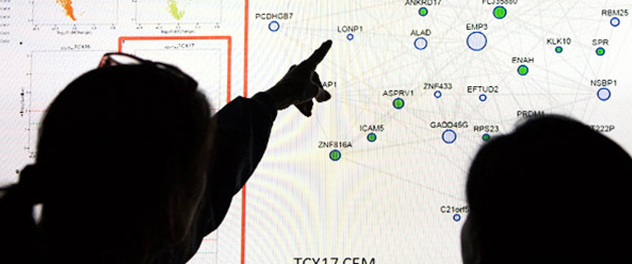
Combining omics data and clinical knowledge
Dr. Ertekin-Taner's lab analyzes large-scale genomic, transcriptomic and epigenomic data on well-characterized patient samples to identify dysregulated genes and pathways in complex neurodegenerative diseases.
Overview
The goal of the Precision Medicine in Neurodegeneration Laboratory of Nilufer Ertekin-Taner, M.D., Ph.D., at Mayo Clinic is to elucidate the complex genetics of Alzheimer's disease by discovering and characterizing genetic factors that influence its risk and modulate biological quantitative phenotypes (endophenotypes), such as gene expression levels and cognition.
Alzheimer's disease is the most common form of dementia, affecting more than 55 million people worldwide. It is characterized primarily by memory decline and impairment in other cognitive areas, including language, executive function and visuospatial abilities. Without effective therapies, the number of people with dementia, including Alzheimer's disease, is expected to exceed 139 million by 2050. It is clear that developing effective therapies for Alzheimer's disease requires a thorough understanding of its pathophysiology, risk and protective factors.
The lab focuses on recognizing the underlying genetic component and identifying genetic risk and protective factors in the disease mechanism. This knowledge could help develop novel therapeutic approaches by identifying druggable targets. Further, genetic risk and protective factors could potentially be used as biomarkers to determine at-risk populations to commence drug therapy in the presymptomatic stage.
Pathologically, Alzheimer's disease is characterized by senile plaques composed predominantly of extracellular accumulation of the amyloid-beta peptide, which is processed from amyloid precursor protein and neurofibrillary tangles. These senile plaques form by intracellular accumulation of the abnormally hyperphosphorylated microtubule-associated protein, tau.
The identification of deterministic mendelian mutations in the amyloid precursor protein, as well as presenilin 1 (PSEN1) and presenilin 2 (PSEN2) genes responsible for early-onset autosomal dominant familial forms of Alzheimer's disease, have led to a better understanding of this disease's pathophysiology. But these mutations explain less than 1% of all cases.
Common cases of late-onset Alzheimer's disease also have a substantial genetic component that is only partially explained by the ε4 allele of the apolipoprotein E gene. This is the only widely accepted genetic risk factor for late-onset Alzheimer's disease. The lab has several approaches that aim to uncover the underlying genetics of late-onset Alzheimer's disease. Learn more about these approaches and the associated research.