Living pharmacy implanted device — EASE project
 Helping patients with inflammatory bowel disease
Helping patients with inflammatory bowel disease
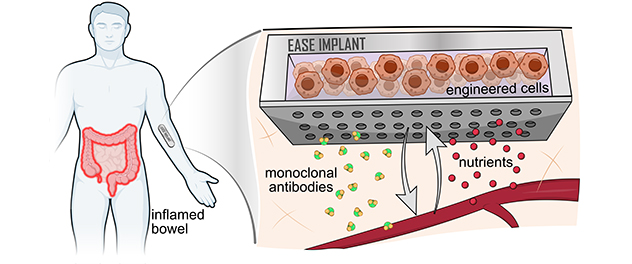
The goal of the EASE project is to develop an implantable device that will deliver anti-inflammatory antibodies to treat patients with inflammatory bowel disease. Also included is a wearable component that will sample sweat to confirm the production of antitumor necrosis factor antibodies and the reduction of inflammatory markers.
The Engage Assess SecretE (EASE) project is a $43 million Advanced Research Projects Agency for Health award to a consortium of five academic institutions and two startups. Mayo Clinic is the lead institution. Dr. Revzin is the principal investigator.
The goal of this project is to develop an implantable device carrying cells that produce anti-inflammatory antibodies to treat inflammatory bowel disease. The implantable device will contain bioelectronic components that can stimulate cells to turn therapy production on and off. This device also will contain a battery and a wireless charging module.
The project began in September 2024 with the hope of a phase 1 clinical trial to be undertaken in 2029. The implantable living pharmacy approach pursued by our team will be paradigm shifting for the field of inflammatory bowel disease treatment. The current standard of care is infusion therapy performed every several weeks in the clinic.