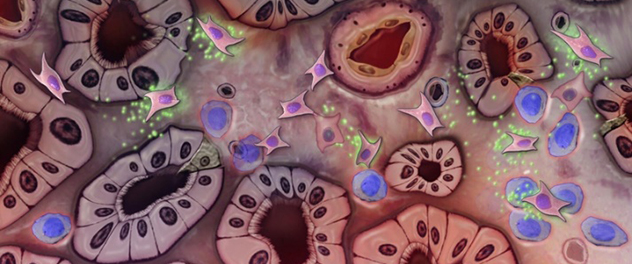
 Potential mesenchymal stem cell activities in an injured kidney
Potential mesenchymal stem cell activities in an injured kidney
Mesenchymal stem cells reaching the kidney may release factors that affect kidney cells and tissues.
Regenerative Nephrology Compassionate Use Program
The U.S. Food and Drug Administration (FDA) has authorized Mayo Clinic to offer mesenchymal stem cells as part of the Regenerative Nephrology Compassionate Use Program. This is an expanded access program. Mesenchymal stem cells are adult stem cells or stromal cells collected from tissues such as bone marrow, fat or umbilical cord (and blood).
Mesenchymal stem cells may be available to:
- Adults with chronic kidney disease. People ages 18 and older with chronic kidney disease may be eligible, but they should not be receiving dialysis. For this program, chronic kidney disease is defined as having an estimated glomerular filtration rate less than 60 mL per minute per 1.73 m2.
- Adults with a kidney transplant. The kidney transplant must be losing function.
People taking part in the program are not involved in a clinical trial. Eligibility criteria may change over time. Contact our program for the latest requirements.
Mesenchymal stem cells are considered investigational. They are not FDA approved to treat kidney disease.
About mesenchymal stem cells and kidney disease
In chronic kidney disease, inflammation in the kidney may cause scarring that furthers loss of kidney function over time, no matter the cause of kidney disease.
Mesenchymal stem cells release factors that may:
- Reduce inflammation in the body and the kidneys.
- Support the health of the remaining kidney cells.
- Slow the processes that cause kidney scarring.
About the program
Mesenchymal stem cells used in the Regenerative Nephrology Compassionate Use Program are culture expanded and manufactured in facilities using Current Good Manufacturing Practices (CGMP). CGMP is the FDA's way of ensuring that manufacturing processes and facilities are properly designed, monitored and controlled.
Program participants will receive around 200 million mesenchymal stem cells. They will be followed for six months to monitor safety.
Cell delivery
Two strategies are being offered to deliver mesenchymal stem cells as part of the program:
- IV delivery. Mesenchymal stem cells may be delivered directly into a vein in the arm using an IV line during a single-day treatment.
- Combination delivery. This strategy involves intra-arterial delivery to each kidney and IV delivery during a two-day treatment. Intra-arterial kidney delivery requires a procedure to enter the arteries in the body.
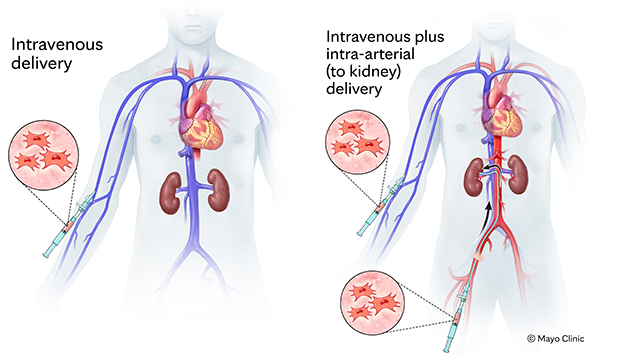 Stem cell delivery options in Mayo Clinic's Regenerative Nephrology Compassionate Use Program
Stem cell delivery options in Mayo Clinic's Regenerative Nephrology Compassionate Use Program
In the program, mesenchymal stem cells are delivered by IV or through a combined approach with both IV and intra-arterial delivery to the arteries of each kidney in a procedure, as shown.
Researchers are still assessing the safety of these delivery routes and how they may influence mesenchymal stem cell actions in a damaged kidney.
Those who are eligible and interested may receive repeat doses of mesenchymal stem cells. They will be followed for safety assessments after each mesenchymal stem cell administration.
Care afterward
Program participants continue to receive regular kidney care with their local nephrology team. Mayo Clinic's Regenerative Nephrology team manages mesenchymal stem cell infusion visits on-site and safety follow-up visits virtually.
Publications
Patel HA, Wang J, Zinn CJ, Learmonth M, Bian X, Jurk D, Lerman LO, Wolfram J, Hickson LJ. Targeting cellular senescence in diabetic kidney disease: Potential of regenerative, cell-based therapies and other senotherapeutic approaches. Kidney International. 2026.
Alatta LS, Elhusseiny KM, Dietz AB, Herrmann SM, Lorenz EC, Lawson DK, Bendel EC, Reisenauer CJ, Misra S, Vaughan LE, Lerman LO, Hickson LJ. Intraarterial autologous mesenchymal stem cell therapy for diabetic kidney disease. Kidney International Reports. 2025.
Patel HA, Wang J, Zinn CJ, Learmonth M, Lerman LO, Wolfram J, Hickson LJ. Fortifying the diabetic kidney disease treatment armamentarium: Multitarget senotherapeutic and regenerative strategies. Journal of the American Society of Nephrology. 2025.
Hickson LJ, Herrmann SM, McNicholas BA, Griffin MD. Progress toward the clinical application of mesenchymal stromal cells and other disease-modulating regenerative therapies: Examples from the field of nephrology. Kidney360. 2021.
Abumoawad A, Saad A, Ferguson CM, Eirin A, Herrmann SM, Hickson LJ, Goksu BB, Bendel E, Misra S, Glockner J, Dietz AB, Lerman LO, Textor SC. In a Phase 1a escalating clinical trial, autologous mesenchymal stem cell infusion for renovascular disease increases blood flow and the glomerular filtration rate while reducing inflammatory biomarkers and blood pressure. Kidney International. 2020.
Saad A, Dietz AB, Herrmann SMS, Hickson LJ, Glockner JF, McKusick MA, Misra S, Bjarnason H, Armstrong AS, Gastineau DA, Lerman LO, Textor SC. Autologous mesenchymal stem cells increase cortical perfusion in renovascular disease. Journal of the American Society of Nephrology. 2017.