Biological mechanisms of neuromuscular disorders
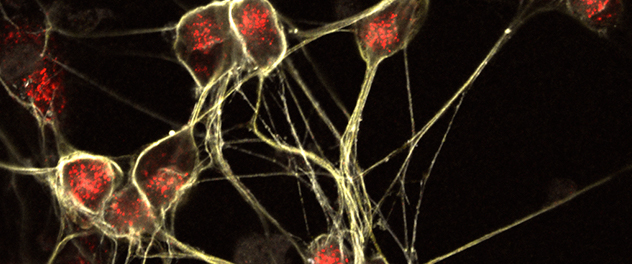
Photographed are human sensory neurons derived from induced pluripotent stem cells.
The Translational Neuromuscular Disease Research Lab is conducting several studies to investigate the basic biological mechanisms of neuromuscular conditions. The goal is to develop treatments and neuroprotection for neuromuscular conditions, with a focus on chemotherapy-induced peripheral neuropathy, spinal muscular atrophy and amyotrophic lateral sclerosis (ALS).
The primary lab model is in vitro human sensory and motor neurons. These neurons are manufactured from human skin biopsy-derived induced pluripotent stem cells. Human neurons allow the lab to take advantage of human biology and study the specific human cells affected in a variety of diseases. The lab banks samples in the Regenerative Medicine Biotrust.
Chemotherapy-induced peripheral neuropathy
Chemotherapy-induced peripheral neuropathy is a painful disorder of the peripheral nervous system that affects more than half of the patients with cancer who are treated with neurotoxic agents such as platinum compounds, vinca alkaloids, taxanes and proteasome inhibitors. As cancer therapies increasingly extend survival and cure rates, the incidence of chemotherapy-induced peripheral neuropathy continues to rise.
To better understand this condition, Dr. Staff's research team developed a human neuronal culture system derived from induced pluripotent stem cells generated from skin biopsies. This system enables a more accurate human-based model of chemotherapy-induced peripheral neuropathy in the lab.
Research in the Translational Neuromuscular Disease Research Lab has identified microtubules and mitochondria as key neuronal components damaged by chemotherapeutic agents. Microtubules, which provide structural support and enable axonal transport, show increased polymerization when exposed to bortezomib — a change also observed with taxanes despite their different anticancer mechanisms. Also, bortezomib disrupts mitochondrial movement along axons, impairing energy delivery within neurons. The lab seeks to define the upstream protein pathways driving these changes to identify targets for neuroprotection.
Spinal muscular atrophy
Spinal muscular atrophy is a genetic neuromuscular disorder caused by deficiency of the survival motor neuron (SMN) protein, most commonly due to mutations or deletions in the SMN1 gene. This deficiency leads to degeneration of lower motor neurons in the spinal cord, resulting in progressive muscle weakness, atrophy and paralysis.
Although spinal muscular atrophy has historically been a leading genetic cause of infant mortality, recent advances in SMN-enhancing therapies have transformed the clinical landscape, shifting the focus toward optimizing outcomes, understanding disease variability, and addressing residual weakness and long-term complications across the lifespan.
In Dr. Staff's lab, research on spinal muscular atrophy centers on understanding motor neuron biology from induced pluripotent stem cell-derived motor neurons from people with spinal muscular atrophy. The lab integrates clinical investigation with translational and lab-based approaches to study disease pathophysiology and identify novel pathomechanisms of disease that may be targets for therapeutic intervention.
ALS
ALS is a progressive and ultimately fatal neurodegenerative disease characterized by the selective loss of upper and lower motor neurons, leading to muscle weakness, paralysis and respiratory failure. Despite advances in genetics and clinical care, the cellular mechanisms that drive motor neuron vulnerability and disease progression remain incompletely understood. Dr. Staff's lab investigates these mechanisms using human induced pluripotent stem cell-derived motor neuron models that enable direct study of ALS in patient-specific, disease-relevant cells.
By reprogramming patient-derived cells into induced pluripotent stem cells and differentiating them into motor neurons, the lab generates experimentally tractable systems that capture key genetic and pathological features of ALS. Using integrated approaches that combine stem cell biology, gene editing, functional genomics, imaging and electrophysiology, the lab defines mutation-specific and convergent disease pathways. The goal is to identify actionable therapeutic targets and translate mechanistic insights into precision strategies that meaningfully alter the course of ALS.
Hereditary peripheral neuropathy (Charcot-Marie-Tooth disease)
Mutations in over 100 genes can lead to peripheral neuropathy. Using human sensory neurons derived from people who have hereditary peripheral neuropathy, also called Charcot-Marie-Tooth disease, Dr. Staff and his research team are investigating how different genetic mutations lead to neuronal damage.