Targeting Mesotrypsin in Breast, Prostate, Pancreatic and Lung Cancers
 Mesotrypsin surface
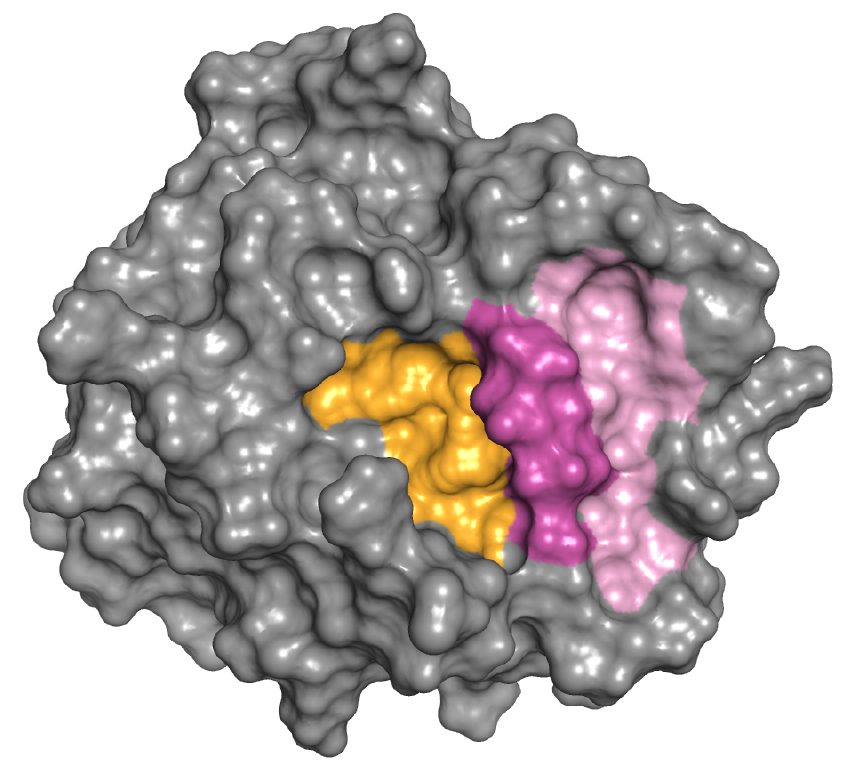
Mesotrypsin surface
This illustration shows that mesotrypsin activity can be blocked by inhibitors that target the active site (yellow) or a unique allosteric site (pink).
Mesotrypsin, encoded by the PRSS3 gene, is a human serine protease whose upregulation has been linked to promoting tumor progression. Mesotrypsin inhibitors could offer valuable tools for deciphering the pathological role of this enzyme. These inhibitors also could form the basis of novel therapeutic strategies targeting mesotrypsin.
Our lab is developing mesotrypsin inhibitors and evaluating their therapeutic potential in preclinical models of multiple cancers.
Mesotrypsin inhibitors could offer a new approach to slowing cancer cell malignant growth, invasion and metastasis.
But targeting mesotrypsin specifically is a challenge because mesotrypsin is one enzyme among a large family of proteases with similar structure and specificity. In addition, mesotrypsin has proved notoriously resistant to inhibition by many inhibitors of other trypsins.
Our lab has created prototype mesotrypsin inhibitors with improved affinity by using site-directed mutagenesis, biochemical assays and X-ray crystallography to define features of mesotrypsin specificity that are distinct from other serine proteases. These engineered inhibitors showed striking anti-cancer activity in physiologically relevant assays of breast cancer cell malignant growth, pancreatic cancer cell invasion and prostate cancer cell invasion.
Our lab has a partnership with the protein engineering laboratory of Niv Papo at Ben-Gurion University of the Negev in Israel. We used directed evolution approaches to screen large libraries of protein inhibitors based on a natural human protease inhibitor called APPI. This collaborative work has identified even more-potent mesotrypsin inhibitors with therapeutic potential.
In other studies, our team used protein crystal structures and in silico docking screens to identify novel small-molecule inhibitors of mesotrypsin. We're now using structure-based approaches to optimize selective small-molecule inhibitors that target a unique allosteric site on the surface of mesotrypsin.
Further development of these polypeptide and small-molecule inhibitors for enhanced mesotrypsin selectivity may generate targeted therapeutics that can reduce tumor progression in people with cancer.