CNS Metabolism and Lipid Physiology
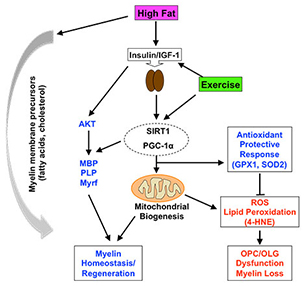
Model depicts the interplay between exercise and dietary fat that is capable of fostering myelin generation in the adult spinal cord.
Diet is an intrinsic aspect of everyday life and is emerging as a major regulator of brain function and plasticity. In particular, the increasing consumption of saturated fats and sugars in a typical Western diet is considered detrimental for central nervous system function. However, based on the high content of lipids in the brain, how to manage consumption of dietary fats for optimal CNS health is controversial.
Myelin is essential to the conduction of nerve impulses in the brain and spinal cord. Myelin loss is a key pathophysiological component of neurological injury and disease, including multiple sclerosis, traumatic brain and spinal cord injury, Alzheimer's disease, stroke, and certain neuropsychiatric disorders. Myelin membranes have a very high lipid-to-protein ratio, in which lipids account for at least 70% of the dry weight. So, myelin assembly requires an extraordinary amount of lipids. This is especially true for lipids such as cholesterol and sphingolipids that are enriched in myelin.
Recent findings from Dr. Scarisbrick's Neuroregeneration and Neurorehabilitation Lab suggest for the first time that consumption of a diet high in saturated fats and sugar compromises the health of myelinating cells. The team also demonstrated that these deleterious effects were prevented by coordinate exercise interventions. These studies have important implications for the design of rehabilitative programs to enhance functional capacity in the brain and spinal cord across the lifespan. Both dietary fat content and exercise are essential considerations in the context of injury and regeneration as well.
Related publications
- Langley MR, Choi CI, Peclat TR, Guo Y, Simon WL, Yoon H, Kleppe L, Lucchinetti CF, Chini CCS, Chini EN, Scarisbrick IA. Critical role of astrocyte NAD+ glycohydrolase in myelin injury and regeneration. Journal of Neuroscience. 2021; doi10.1523/JNEUROSCI.2264-20.2021.
- Triplet EM, Scarisbrick IA. Statin use is associated with reduced motor recovery after spinal cord injury. Spinal Cord Series and Cases. 2021; doi:10.1038/s41394-020-00378-y.
- Kim HN, Langley MR, Simon WL, Yoon H, Kleppe L, Lanza IR, LeBrasseur NK, Matveyenko A, Scarisbrick IA. A Western diet impairs CNS energy homeostasis and recovery after spinal cord injury: Link to astrocyte metabolism. Neurobiology of Disease. 2020; doi:10.1016/j.nbd.2020.104934
- Langley MR, Choi C-I, Peclat TR, Simon W, Yoon H, Kleppe L, Chini CCS, Chini EN, Scarisbrick IA. CD38 dependent NAD+ depletion contributes to oligodendrocyte loss and inhibition of myelin regeneration. bioRxiv. 2020; doi:10.1101/2020.06.10.143941.
- Langley MR, Triplet EM, Scarisbrick IA. Dietary influence on central nervous system myelin production, injury and regeneration. Biochimica Biophysica Acta — Molecular Basis of Disease. 2020; doi:10.1016/j.bbadis.2020.165779.
- Langley MR, Yoon H, Kim HN, Choi CI, Simon W, Kleppe L, Lanza IR, LeBrasseur NK, Matveyenko A, Scarisbrick IA. High fat diet consumption results in mitochondrial dysfunction, oxidative stress and oligodendrocyte loss in the central nervous system. Biochimica Biophysica Acta — Molecular Basis of Disease. 2020; 10.1016/j.bbadis.2019.165630.
- Yoon H, Kleven A, Paulsen A, Kleppe L, Wu J, Ying Z, Gomez-Pinilla F, Scarisbrick IA. Interplay between exercise and dietary fat modulates myelinogenesis in the central nervous system. Biochimica et Biophysica Acta — Molecular Basis of Disease. 2016; doi:10.1016/j.bbadis.2016.01.019.