Research
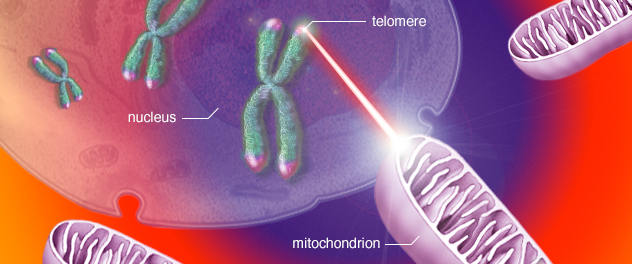 Mitochondrial stress drives telomere damage and senescence
Mitochondrial stress drives telomere damage and senescence
Dr. Passos' research has demonstrated that telomeres are preferential targets of mitochondrial-derived oxidative stress, linking mitochondrial dysfunction to telomere damage and cellular senescence.
Telomere damage as a biomarker of aging
Telomeres are key drivers of cellular senescence. When damaged or critically short, telomeres trigger a persistent DNA damage response that promotes stable cell-cycle arrest and the development of senescence.
Dr. Passos' research has further revealed a direct mechanistic link between mitochondria and telomere dysfunction. His work demonstrated that mitochondrial-derived oxidative stress preferentially targets telomeres, coupling mitochondrial dysfunction to telomere damage and the initiation of senescence pathways. This mitochondria-telomere axis provides an important framework for understanding how metabolic and stress signals accelerate biological aging.
Dr. Passos' team has shown that telomere-associated DNA damage foci (TAF), defined by the co-localization of DNA damage response proteins with telomeric regions, increase with aging across multiple tissues, including skin, liver, intestine, lung, heart, skeletal muscle, brain and bone. Elevated TAF levels also have been observed in human diseases, such as chronic obstructive pulmonary disease, bronchiectasis, idiopathic pulmonary fibrosis, nonalcoholic and alcoholic liver disease, and myocardial infarction. In addition, Dr. Passos' team has shown that processes known to accelerate aging, including chronic inflammation, obesity, mitochondrial dysfunction and impaired autophagy, promote TAF formation.
Based on extensive experimental evidence accumulated over more than a decade, Dr. Passos' research proposes that TAF represents one of the most robust and reliable markers of cellular senescence in vivo. The lab is now advancing the translational application of TAF as a potential prognostic biomarker to assess biological age and monitor the effectiveness of interventions targeting aging and age-related disease.
Related publications
- Rossiello F, Jurk D, Passos JF, d'Adda di Fagagna F. Telomere dysfunction in ageing and age-related diseases. Nature Cell Biology. 2022.
- Lagnado A, Leslie J, Ruchaud-Sparagano MH, Victorelli S, Hirsova P, Ogrodnik M, Collins AL, Vizioli MG, Habiballa L, Saretzki G, Evans SA, Salmonowicz H, Hruby A, Geh D, Pavelko KD, Dolan D, Reeves HL, Grellscheid S, Wilson CH, Pandanaboyana S, Doolittle M, von Zglinicki T, Oakley F, Gallage S, Wilson CL, Birch J, Carroll B, Chapman J, Heikenwalder M, Neretti N, Khosla S, Masuda CA, Tchkonia T, Kirkland JL, Jurk D, Mann DA, Passos JF. Neutrophils induce paracrine telomere dysfunction and senescence in ROS-dependent manner. The EMBO Journal. 2021.
- Anderson R, Lagnado A, Maggiorani D, Walaszczyk A, Dookun E, Chapman J, Birch J, Salmonowicz H, Ognodnik M, Jurk D, Proctor C, Correia-Mela C, Victorelli S, Fielder E, Berlinguer-Palmini R, Owens A, Greaves LC, Kolsky KL, Parini A, Douin-Echinar V, LeBrasseur NK, Arthur HM, Tual-Chalot S, Schafer MJ, Roos CM, Miller JD, Robertson N, Mann J, Adams PD, Tchkonia T, Kirkland JL, Mialet-Perez J, Richardson GD, Passos JF. Length-independent telomere damage drives post-mitotic cardiomyocyte senescence. The EMBO Journal. 2019.
- Hewitt G, Jurk D, Marques F, Correia-Melo C, Hardy T, Gackowska A, Anderson R, Taschuk M, Mann J, Passos JF. Telomeres are favoured targets of a persistent DNA damage response in ageing and stress-induced senescence. Nature Communications. 2012.
State-of-the-art methodology
To accurately measure and visualize telomere dysfunction in multiple tissues, the Cell and Molecular Aging Lab has optimized different methods using both conventional fluorescent microscopy and super-resolution microscopy. Also, Dr. Passos' lab developed computer imaging software to quantify telomere dysfunction in different cells and tissues in an unbiased and reliable manner.
Dr. Passos and his team continue to work toward developing novel methods and analytical tools to investigate telomeres and senescence in tissues.
Mitochondria and aging
The Cell and Molecular Aging Lab's research shows that mitochondria play a central role in senescent cells and the biological processes that drive aging and age-related disease. The lab focuses on defining the molecular mechanisms that connect mitochondrial stress responses to cellular senescence and tissue dysfunction.
Key discoveries from the lab demonstrated that mitochondria are critical regulators of the senescence-associated secretory phenotype (SASP), a major mediator of chronic inflammation during aging. The lab subsequently identified mitochondrial sub-lethal apoptosis as a mechanism that promotes intracellular mitochondrial DNA leakage and downstream activation of the SASP. More recently, the group showed that mitochondrial RNA released from senescent cells engages RNA-sensing innate immune pathways that sustain inflammatory SASP signaling.
Together, these findings establish mitochondria as fundamental drivers of senescence-associated inflammation and highlight mitochondrial stress pathways as promising therapeutic targets to promote healthier aging.
Related publications
- Victorelli S, Eppard M, Woo SH, Everts SPA, Martini H, Pirius N, Franco AC, Han Y, Saul D, Splinter PL, O'Hara SP, Valenzuela-Pérez L, Lee HSK, Jurk D, LaRusso NF, Hirsova P, Passos JF. Mitochondrial RNA cytosolic leakage drives the SASP. Nature Communications. 2025.
- Victorelli S, Salmonowicz H, Chapman J, Martini H, Vizioli MG, Riley JS, Cloix C, Hall-Younger E, Machado Espindola-Netto J, Jurk D, Lagnado AB, Sales Gomez L, Farr JN, Saul D, Reed R, Kelly G, Eppard M, Greaves LC, Dou Z, Pirius N, Szczepanowska K, Porritt RA, Huang H, Huang TY, Mann DA, Masuda CA, Khosla S, Dai H, Kaufmann SH, Zacharioudakis E, Gavathiotis E, LeBrasseur NK, Lei X, Sainz AG, Korolchuk VI, Adams PD, Shadel GS, Tait SWG, Passos JF. Apoptotic stress causes mtDNA release during senescence and drives the SASP. Nature. 2023.
- Correia-Melo C, Marques F, Anderson R, Hewitt G, Hewitt R, Cole J, Carroll BM, Miwa S, Birch J, Merz A, Rushton MD, Charles M, Jurk D, Tait S, Czapiewski R, Greaves, L Nelson G, Bohlooly-Y M, Rodriguez-Cuenca S, Vidal-Puig A, Mann D, Saretzki G, Quarato G, Green DR, Adams PD, von Zglinicki T, Korolchuk VI, Passos JF. Mitochondria are required for pro-ageing features of the senescent phenotype. The EMBO Journal. 2016.
Dr. Passos' research team is investigating the molecular processes by which mitochondria drive senescence. The team hopes to find new therapies to improve health during old age. The team also is conducting drug screens targeting mitochondrial function to find new therapies to counteract the aging process.