-
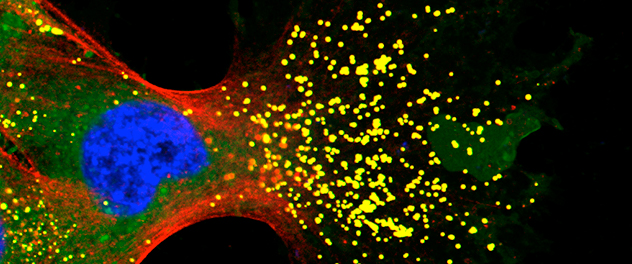
Metabolic changes that fuel metastasis
Stored lipids can promote tumor cell migration and invasion. Our lab is investigating how these pathways are aberrantly activated in pancreatic cancer, leading to increased invasive migration. This image shows a fluorescent micrograph of a pancreatic cancer cell extending a large migratory protrusion, with actin in red, lipid droplets in yellow and the nucleus in blue.
-
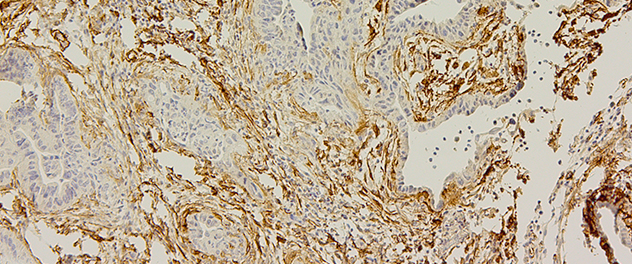
The complex tumor microenvironment
Pancreatic tumors include ductal tumor cells surrounded by an activated stroma. We're studying how inflammatory signals activate the metastatic machinery. This image shows a tissue section of a pancreatic tumor stained using immunohistochemistry, with a brown stain showing an extensive stromal component surrounding the ductal tumor cells.
-
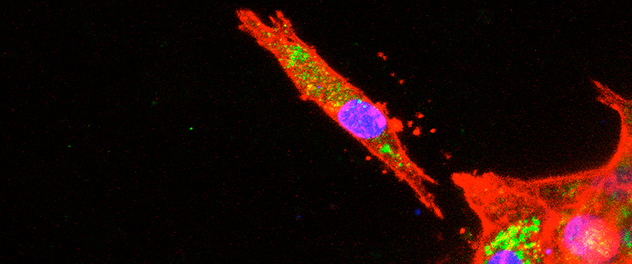
A model of cancer metastasis
Pancreatic cancer cells are grown in a 3D environment. Invasive migration away from the spheroid can model metastatic invasion in vitro. We’re investigating the mechanisms by which tumor cells alter their metabolism to drive metastasis. This image shows a fluorescent micrograph of pancreatic tumor cells grown in a 3D extracellular matrix, stained for actin in red, highlighting one cell invading away from the spheroid.
Overview
The Cell Biology of Metastasis Laboratory investigates the mechanisms by which cancer spreads throughout the body.
Most cancer deaths are associated with metastasis, but current cancer therapies don't inhibit the metastatic process. In the Cell Biology of Metastasis Lab, principal investigator Gina Razidlo, Ph.D., and her research team hope to overcome that challenge and improve survival.
We're using biochemistry and cell biology to study tumor cell migration and invasion, with the prediction that targeted inhibition of metastatic invasion in combination with therapies to target the primary tumor will improve outcomes.
Focus areas
Our lab has two main focus areas of research:
Signaling pathways that regulate cytoskeletal remodeling
Cell migration is driven by remodeling of the dynamic actin cytoskeleton. This is coupled to upstream signaling events driven by receptor signaling and interactions with the extracellular matrix. Signaling through the small GTPases Rac, Cdc42 and Rho leads to changes in actin dynamics.
We’re investigating how these pathways are aberrantly activated in pancreatic cancer, leading to increased invasive migration.
Signals from the tumor microenvironment
The complex tumor microenvironment provides cues that signal to tumor cells and promote tumor growth and metastasis. Dr. Razidlo is particularly interested in how both inflammation and excess lipids contribute to metastasis.
First, inflammation is a risk factor for pancreatic cancer development and contributes to increased tumor progression and metastasis in multiple cancer models. Our lab is studying how inflammatory signals activate the metastatic machinery. Second, invasive migration is a highly energy-intensive process and requires generating sufficient ATP to power cytoskeletal and membrane remodeling. We're investigating the mechanisms by which tumor cells alter their metabolism to use excess lipids to drive metastasis.