-
Supporting a premier gene, virus and cell therapy program
A multidisciplinary team of scientists and physicians in the Department of Molecular Medicine work together to realize the full potential of virus, gene and cell therapies from basic science discovery to clinical translation.
-

Using the measles virus to attack cancer cells
The measles virus spreads from primary human monocyte-derived macrophages to primary human airway epithelial cells. Copyright © 2016, American Society for Microbiology.
-
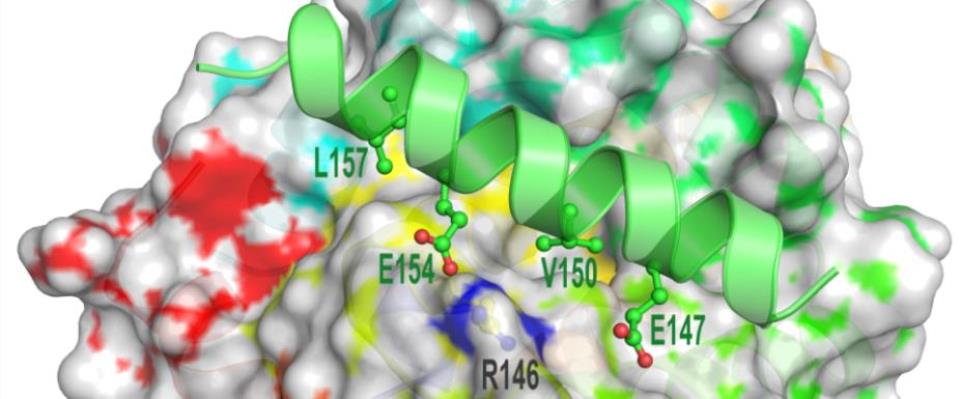
Targeting cancer on a molecular level
The Department of Molecular Medicine at Mayo Clinic is developing a premier virus, gene and cell therapy program, bringing promising therapeutics from bench to bedside in a timely manner.
Overview
The Department of Molecular Medicine is engaged in research and education to build a premier virus, gene and cell therapy program and translate promising therapeutics from bench to bedside in a timely manner. The department discovers, teaches and translates virus, cell and gene therapies to strategically position Mayo Clinic as a premier center providing hope for patients around the globe.
The wide range of disciplines represented by the Department of Molecular Medicine's leadership team and staff facilitates a breadth of translational activities. The researchers in the Department of Molecular Medicine are in the unique position of being able to move a basic science discovery all the way through to clinical trials and the development of novel therapies, all within the department and in an expedited fashion. For instance, instead of the standard 5- to 7-year industry time frame, the department has moved its attenuated measles virus therapy for ovarian cancer from discovery to clinical trials within three years. This integrated process enables the advancement of science and the quick delivery of new treatments to patients.
Research focus areas
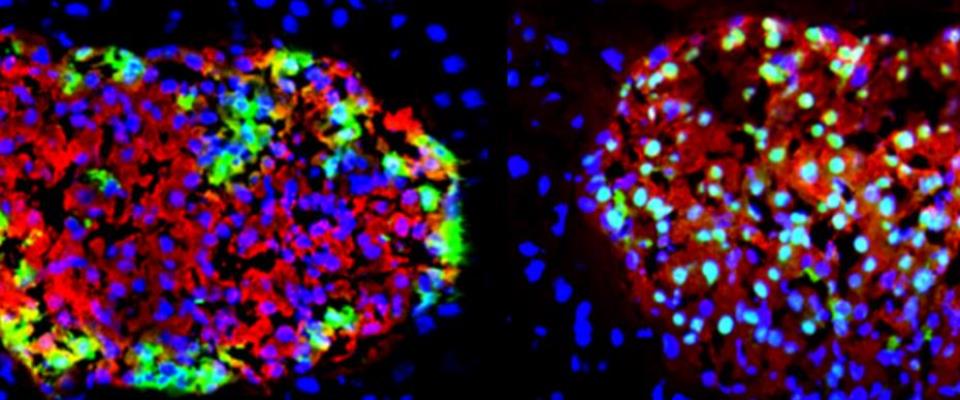
Researchers from the Department of Molecular Medicine help personalize each person's treatment for optimal care. The department's research team focuses on advancing research into biomarker discovery. Researchers also are involved in applying their expertise in gene therapy to the field of regenerative medicine, particularly in the areas of beta cell regeneration and liver regeneration.
The Department of Molecular Medicine works with the Novel Therapeutics and Therapeutic Modalities Research Program within Mayo Clinic Comprehensive Cancer Center — Research to develop new gene delivery systems and gene- and virus-based therapies for cancer treatment. The program conducts research on all Mayo Clinic campuses.
Core facilities such as the Toxicology and Pharmacology Shared Resource manufacture clinical-grade engineered viruses and perform preclinical toxicology and biodistribution studies to support the department's clinical trials.
Affiliations