 Acellular therapies
Acellular therapies
Acellular therapy projects in the Mayo Clinic Van Cleve Cardiac Regenerative Medicine Program include a biologically potentiated matrix to heal and repair damaged tissue, a shelf-stable product to facilitate quick use of biologics-based therapies for heart attacks, a minimally invasive therapy for patients with heart failure, and a virus-free gene therapy to help prevent sudden cardiac death.
Acellular Therapies
Cell-free regeneration
Exosome microscopy
Exosome microscopy
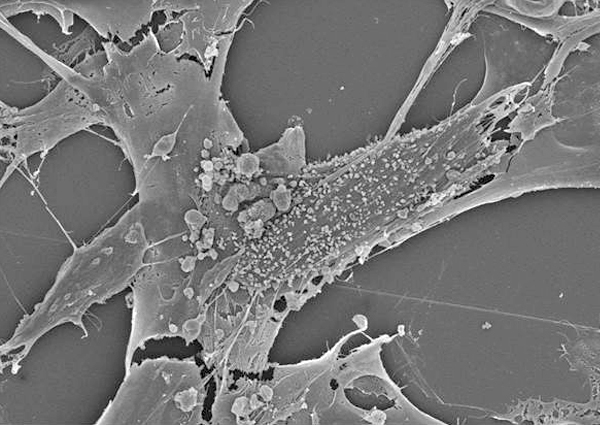
Exosome microscopy
Scanning electron microscope images of extracellular vesicles being shed from mesenchymal stem cells. Images were created in the laboratory of Atta Behfar, M.D., Ph.D., at Mayo Clinic.
Stem cells work by serving as reservoirs that release molecules capable of inducing repair within the ailing heart. Analyzing the regenerative signals secreted by stem cells offers the prospect of realizing cell-free (acellular) therapy. By eliminating cells from regenerative medicine therapies, the therapies can be stored on the shelf and be readily available for use. These "off-the-shelf" technologies improve consistency and accessibility and lower costs.
Exosome microscopy
Exosome microscopy
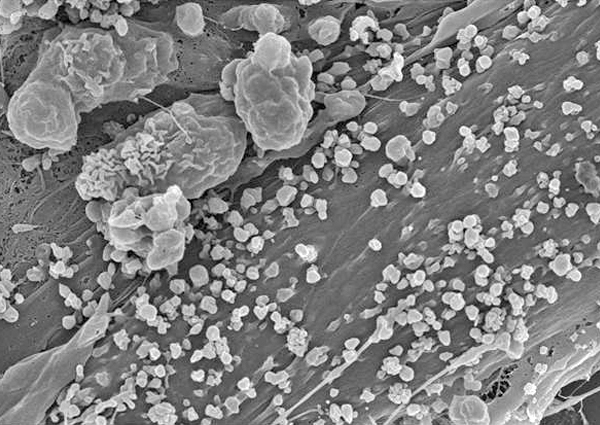
Exosome microscopy
Scanning electron microscope images of extracellular vesicles being shed from mesenchymal stem cells. Images were created in the laboratory of Atta Behfar, M.D., Ph.D., at Mayo Clinic.
Research background
The discovery of extracellular vesicles (also called exosomes) has revolutionized the way researchers view regenerative medicine. Several initiatives in the Mayo Clinic Van Cleve Cardiac Regenerative Medicine Program are accelerating the use of extracellular vesicles in clinical practice.
- High-potency extracellular vesicles. Deriving and preserving exosomes in sufficient abundance to ensure a regenerative benefit presents a challenge. In partnership with the Center for Regenerative Biotherapeutics, the Mayo Clinic Van Cleve Cardiac Regenerative Medicine Program has created a manufacturing protocol that achieves this purpose. The derived off-the-shelf exosome platform facilitates the use of this new technology in many areas of medicine and surgery.
- Engineering extracellular vesicles. To achieve a targeted therapeutic outcome, the program is utilizing gene, cellular and environmental engineering to tailor the regenerative potency of exosomes. This approach has uncovered a new science for inducement of tissue healing and has paved the way toward virus-free gene therapy.
Projects
Exosome functions
Exosome functions
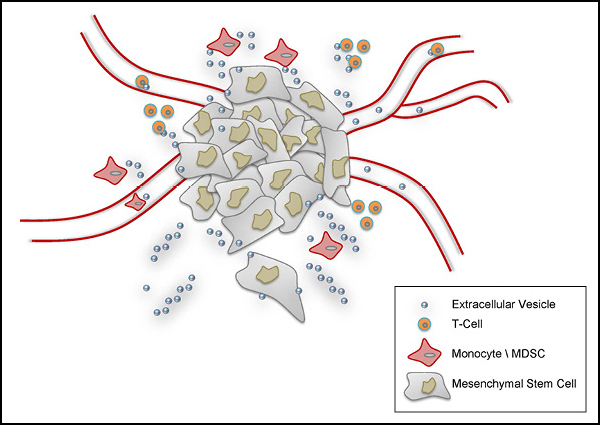
Exosome functions
Extracellular vesicles shed by mesenchymal stem cells participate in many cellular functions, such as angiogenesis, cell proliferation and migration, immunosuppression, and wound healing.
Investigators in the Mayo Clinic Van Cleve Cardiac Regenerative Medicine Program are actively pursuing acellular therapy projects in various stages of discovery, translation and application. Research projects include:
-
Biologically potentiated matrix. Researchers in the Mayo Clinic Van Cleve Cardiac Regenerative Medicine Program are developing an off-the-shelf regenerative medicine product to heal and repair damaged tissue. By combining exosomes with natural matrix proteins from the body, they have developed a biologically enhanced gel.
Investigators are now actively testing this technological platform to restore blood flow in peripheral vascular disease and wound healing. Clinical trials to test this novel regenerative tool are planned for 2020.
-
Exosome-based regeneration in heart attack (myocardial infarction). The American Heart Association reports that acute heart attacks occur once every 36 seconds in the United States, affecting 1 million individuals on an annual basis.
Cell-based regenerative technologies have historically been poorly compatible with acute syndromes due to the significant time lag required to prepare a dose. Establishment of a room temperature-stable product that can be left on the shelf in a surgical or procedure room would allow the use of biologics-based therapies at the exact time they are needed.
The Mayo Clinic Van Cleve Cardiac Regenerative Medicine Program — through optimization of exosome science — has established this platform and is poised to start clinical testing in 2019.
-
Heart failure. Cell-based therapies have been tested in the treatment of cardiovascular disease since the early 2000s. This clinical experience has allowed for many new clinical and mechanistic observations, facilitating advances in regenerative science in the treatment of heart failure.
The Mayo Clinic Van Cleve Cardiac Regenerative Medicine Program has established a novel extracellular vesicle to restore small blood vessel content, reduce scar size and dampen inflammatory signals in patients with failing hearts. Taking advantage of the small size of this new exosome, program researchers have developed a minimally invasive delivery approach that would allow for repeat dosing.
-
Virus-free gene therapy. Existing treatment options cannot prevent the thousands of cases of sudden cardiac deaths each year in the United States. Researchers in the Mayo Clinic Van Cleve Cardiac Regenerative Medicine Program are working to develop an alternative, more effective treatment to target and genetically modify the cell populations responsible for ventricular fibrillation.
To address this unmet need in cardiovascular disease, the program focuses on approaches that would deliver healthy genes to the heart in syndromes such as cardiac arrhythmias, which are resistant to treatments in the current standard of care. Breakthroughs in DNA and RNA gene engineering and encapsulation have allowed the program to develop a new approach to deliver gene therapy without the need for viruses.